 
Printf("I'm sorry,but you have enter an inavalid selection. Printf("The program will be terminated\n\n") Printf("Thank you for using this program!!\n") 
Printf("Are you sure you want to exit? (Y/N): ") SetConsoleTextAttribute (GetStdHandle (STD_OUTPUT_HANDLE), FOREGROUND_RED) Printf("Verify your guess by typing it one more time\n") Printf("Try to guess the jackpot number \nbetweern 1 and 10!\n") With a closed-end manometer, no change would be observed, since the vaporized liquid would contribute equal, opposing pressures in both. The answers are as follows: 762 711 51 torr drop 13. Use 1 atm 101.325 kPa in the second part. Printf("Would you want to continue? (Y/N): ") Convert 615 mm Hg to atmospheres using 760 mm Hg 1 atm. Printf("I'm sorry,but you have enter an inavalid selection.Please try again") Atmospheres : The standard atmospheric (symbol: atm) is a unit of pressure which is a non-SI units. It is widely used, and its value is approximately equal to a pressure of 1 torr. Printf("Are you sure you want to exit? (1/0): ") Millimeter Mercury (0☌) : Millimeter of mercury is a small pressure unit which represents the pressure pushing down due to gravity of any volume of liquid mercury which is 1mm high. Printf("Number of mol -> %f",number_of_mol) Volume=(float)(number_of_mol*0.082057*temperature)/pressure 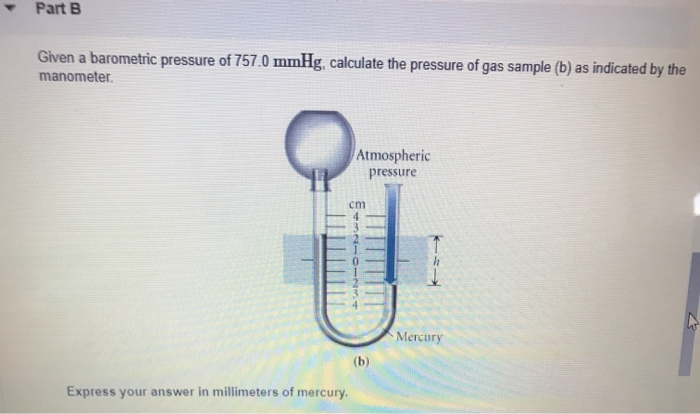
1 psi 6,894.757 Pa: millimeters of mercury: mmHg: Non-SI (International) 1 mmHg 133.3224 Pa: bar: bar: Non-SI (International) 1 bar 100,000 Pa: torr: Torr. Our conversions provide a quick and easy way to convert between Pressure units. Pressure=(float)(number_of_mol*0.082057*temperature)/volume Printf("Please key in the temperature:") Atmospheric pressure is the force exerted by the Earth’s atmosphere on a. This measurement is commonly used to represent atmospheric pressure. 1 mmHg 0.0013157895567935 Atm mmHg is an abbreviation of millimeters of Mercury Atm is an abbreviation of Atmosphere. Printf("Please key in the number of mol:\n") In scientific and engineering contexts, the measurement 760 mm of Hg or 760 mm of water refers to the pressure exerted by a column of mercury (Hg) or water that is 760 millimeters in height. To convert pressure from mmHg to atm, you can use the following formula: atmmmHg760.0atm760. Printf("+WELCOME TO CHEMISTRY QUIZ WORLD+\n") Printf("Do you want to start now(Y/N) ") Printf("\n\nThis is a CHEMISTRY QUIZ\n") SetConsoleTextAttribute(GetStdHandle(STD_OUTPUT_HANDLE), BACKGROUND_RED BACKGROUND_INTENSITY) SetConsoleTextAttribute (GetStdHandle (STD_OUTPUT_HANDLE), FOREGROUND_BLUE) 0.98947 Atmospheres (atm) Visit 752 Atmospheres to mmHg Conversion Millimeter Mercury (0☌) : Millimeter of mercury is a small pressure unit which represents the pressure pushing down due to gravity of any volume of liquid mercury which is 1mm high. This is a chem quiz that contain 20 question but i need to random everytime 10 question,other than this,also have a calculator,& mini games #includeįloat pressure,volume,number_of_mol,temperature Ĭhar answer1,answer2,answer3,answer4,answer5,answer6,answer7,answer8,answer9,answer10,answer11,answer12,answer13,answer14,answer15,answer16,answer17,answer18,answer19,answer20 The barometric pressure pushing down on the liquid in the glass then forces the liquid up the straw.I got no idea how to correct the error,can help me? We can use a drinking straw because sucking on it removes air and thereby reduces the pressure inside the straw. 
We make use of barometric pressure in many ways. Instead, what we notice are changes in the pressure, such as when our ears pop in fast elevators in skyscrapers or in airplanes during rapid changes in altitude. We are so accustomed to living under this pressure that we never notice it. It is defined as the ratio of 760 to 1standard atmosphere, approximately equals one millimeter of mercury. 0.99342 Atmospheres (atm) Visit 755 Atmospheres to mmHg Conversion Millimeter Mercury (0☌) : Millimeter of mercury is a small pressure unit which represents the pressure pushing down due to gravity of any volume of liquid mercury which is 1mm high. Torr : The torr (the symbol: Torr) is a unit of measurement for pressure, but it is a non-SI unit. Thus a pressure of 1 atm equals 760 mmHg exactly. It is widely used, and its value is approximately equal to a pressure of 1 torr. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
